A proprietary, innovative technology from Leoni gives plastic surfaces a germ-killing effect. The process is based on the Lewis acid-base theory and provides a long-term hygiene benefit for hospitals and doctors’ practices by minimizing the risk of infection with antimicrobial cables, cable systems and devices.
Microorganisms continue to cause infections in the healthcare industry, where countless patients come into contact with multiresistant bacteria. The Centers for Disease Control and Prevention (CDC) estimates “that nearly 100,000 people die each year due to hospital-acquired infections” within the USA alone.[1] Young children and older or vulnerable people are at particular risk.
These infections can occur because of gaps in hygiene as well as the fact that bacteria are becoming more resistant and aggressive. A recent focus has been placed on such medical equipment as ECG, ultrasound, catheters and endoscopes as well as their cables. For instance, one study found that more than 77% of ECG cables are colonized by antibiotic-resistant pathogens[2].
Acid-based process
The risk of infection can be substantially reduced through the use of antimicrobial cables, cable systems and equipment casings. LEONI’s process gives the treated objects a special property that resembles the principles of human skin’s protective shield of acids. A significant decrease in bacteria is already evident on the surfaces after a short time. LEONI’s process, which is based on the Lewis acid-base theory, imitates this effect for plastic surfaces. It involves acid ions being released on the surface of cables that lower the pH value on the outer surface of the jacket. This restricts the cell functions of the bacteria and their separation, so that they ultimately die. This is made possible by firmly incorporating a special metal oxide in the plastic matrix of the cable jacket. It can be added in variable doses, with even a low concentration showing more than 99.99% bacteria-killing effectiveness. The mechanical properties of the cable and its handling remain unchanged.
Testing verifies the process
The effect is microbiologically verified, with the evidence provided by an accredited laboratory and independent hospital hygiene experts. The test involved the most well known and most frequently occurring hospital bacteria as well as germs that have developed resistance—specifically by applying various incubation periods and concentrations. The test specimen, an approximately 4-cm (~ 1.57-in.) long section of cable, is incubated with 109 CFU*/ml for six hours. Thereafter the specimen is wound across a blood agar plate, which is incubated at 37°C (98.6°F), every three hours. The findings correspond to the results of tests in practice, involving various timeframes and concentrations on the surface. A significantly faster reduction is evident in the case of a minor bacterial load. This method describes not only the biostatic and biocide effect in planktonic condition, but also assesses the adherence and the biofilm formation of the bacteria. Blocking these is the key to eliminating germs on the surface.
LEONI’s antimicrobial cables have also passed initial tests in accordance with DIN EN ISO 10993-5 and DIN EN ISO 10993-10 for medical equipment.
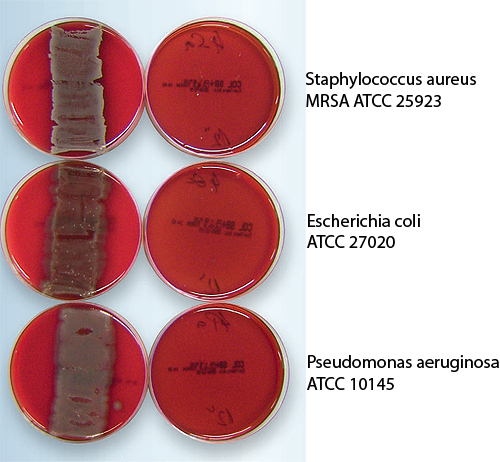
The key benefit is that this acid technology maintains the hygienic effect. Another benefit is that the pathogens are destroyed not from the inside, but from the outside across the cell envelope; the formation of biofilm on the cable or the device casing is thereby stemmed, making it more difficult for bacteria to colonize the surface. The median grain size of the metal oxide firmly incorporated into the plastic is >1 μm.
Equipping device casings, cables and leads with this new type of antimicrobial technology can make a key contribution to closing unwanted gaps in the hygiene chain.
LEONI, Business Unit Healthcare
www.leoni-healthcare.com
Footnotes
[1] www.safepatientproject.org: http://safepatientproject.org/press_release/cdc-hospital-infection-report-shows-more-effort-needed; (12/02/2014)
[2] Donna Quinton Brown; ElectrocardiographyWires: A Potential Source of Infection; NTI News Section B; 106 (2006)
* CFU = colony-forming units



Leave a Reply
You must be logged in to post a comment.